Submitted by Anna Davies on Fri, 01/02/2013 - 11:39
A collaboration between scientists in Cambridge, Oxford and New York has determined the structure of a crucial Trypanosome receptor. The findings, published recently in Proceedings of the National Academy of Sciences have revealed the structure of a crucial receptor found on the surface of trypanosome.
 African Trypanosomes are vector borne parasites that can cause severe disease in humans and animals in sub-Saharan Africa. However, not all trypanosomes cause infections in humans. As modern primates arose in the Rift Valley, they developed an innate immunity which protected them from trypanosomes, since when there has been an evolutionary arms race between trypanosomes and primates. To date, the vast majority of Trypanosomes cannot infect primates, but a few have evolved molecular mechanisms to infect humans, such as T. brucei gambiense.
African Trypanosomes are vector borne parasites that can cause severe disease in humans and animals in sub-Saharan Africa. However, not all trypanosomes cause infections in humans. As modern primates arose in the Rift Valley, they developed an innate immunity which protected them from trypanosomes, since when there has been an evolutionary arms race between trypanosomes and primates. To date, the vast majority of Trypanosomes cannot infect primates, but a few have evolved molecular mechanisms to infect humans, such as T. brucei gambiense.
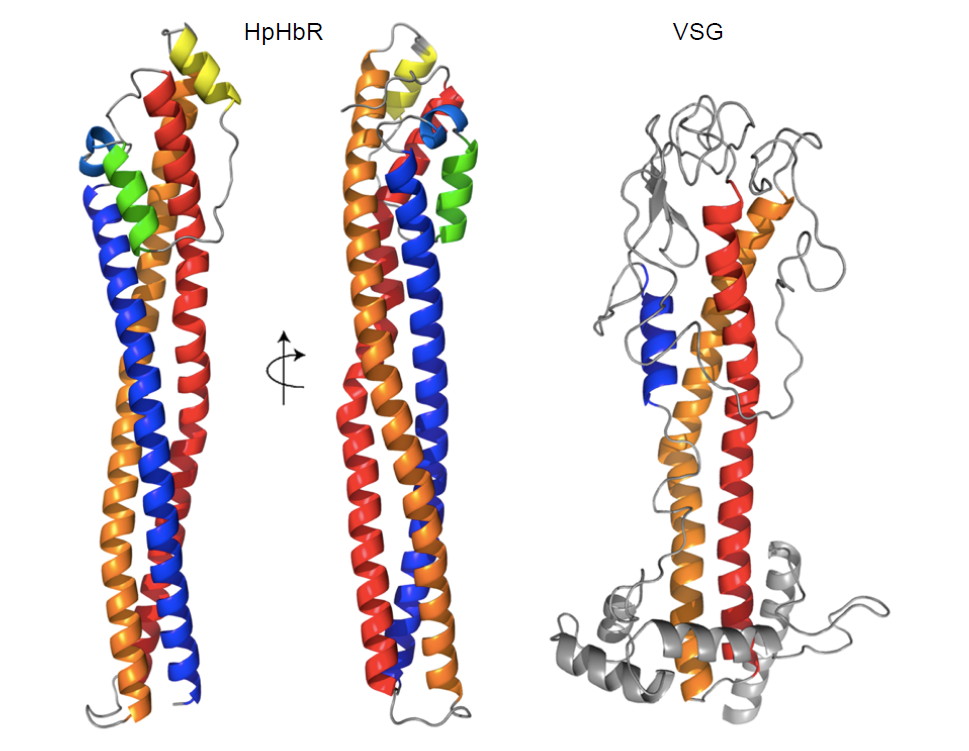
African trypanosomes are protected by a densely packed surface monolayer of variant surface glycoprotein, or VSG. The haptoglobin-hemoglobin receptor (HpHbR) within this VSG coat is used by the trypanosome for haem acquisition. The receptor is exploited by a human innate immunity molecule, Trypanolytic Factor 1 (TLF1), which enters the trypanosome by binding the HpHbR. A collaboration between Professor Mark Carrington’s lab and those of Matt Higgins in Oxford and Jayne Raper in New York has determined the structure of HpHbR, revealing an elongated three α-helical bundle with a small membrane distal head (see Fig 1). Mapping of the HpHb-binding site has shown that a single HbHpR polymorphism unique to human infective T. brucei gambiense is sufficient to reduce binding of both HpHb and TLF1, modulating ligand affinity in a delicate balancing act that allows the Trypanosome to acquire haem but avoids uptake of the lethal TLF1.

